EPD-2520 Pipeline
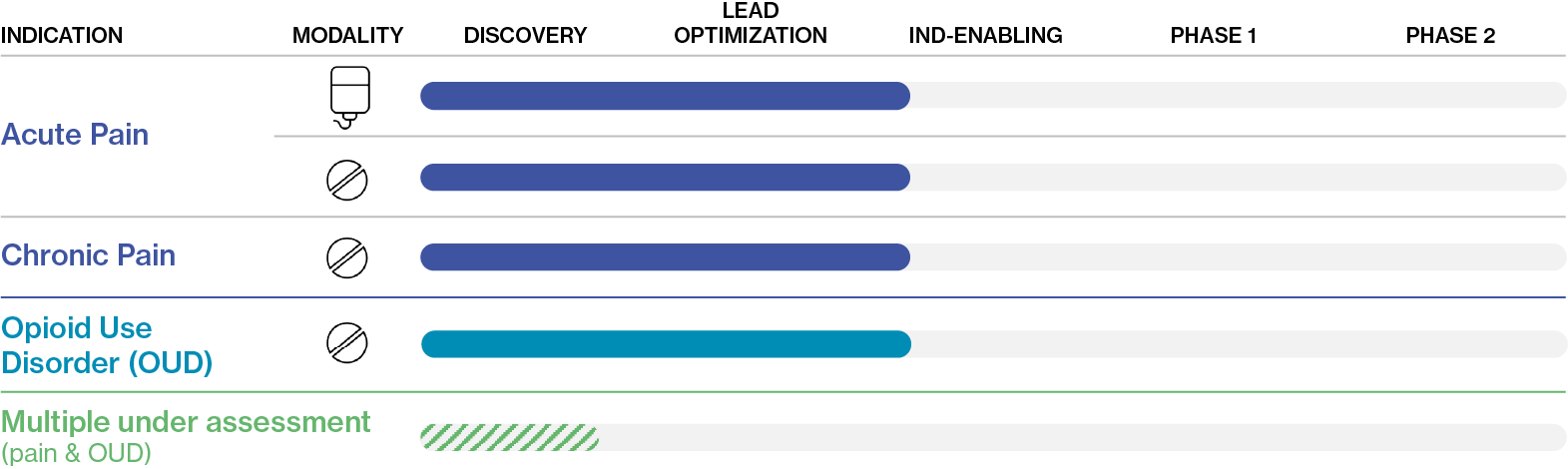
Our upcoming Phase 1 clinical trials for the pain indications and Opioid Use Disorder (OUD) are a single study, designed to provide clinical proof of concept of the key risks for an opioid:
- Respiratory depression
- Analgesia
- Addictive liability in a study in recreational opioid users



